- Blog
- Linked list stack c
- Qcast download
- Does windscribe vpn work with netflix
- Macaroni salad recipes
- Virtual families 2 android cheats
- Cortas splatformer
- Toolbox for pages pirate bay
- Mini calendar 2015
- Cathode ray experiment faraday
- Upload mudic to vesa
- Use microsoft remote desktop
- Fl studio producer edition free download 12-5
- Maxqda separate code segments
- Play nebulous online free
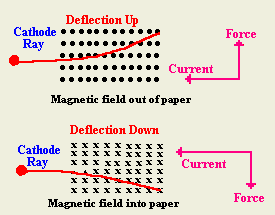
Joseph John (J.J.) Thomson first becomes interested in the discharge of electricity through a gas a low pressure, that is to say, cathode rays.ġ892 Heinrich Hertz who has concluded (incorrectly) that cathode rays must be some form of wave, shows that the rays can penetrate thin foils of metal, which he takes to support the wave hypothesis. In 1891, he changes the name to "electron."ġ876 Eugen Goldstein shows that the radiation in a vacuum tube produced when an electric current is forced through the tube starts at the cathode Goldstein introduces the term cathode ray to describe the light emitted.ġ881 Herman Ludwig von Helmholtz shows that the electrical charges in atoms are divided into definite integral portions, suggesting the idea that there is a smallest unit of electricity.ġ883 Heinrich Hertz shows that cathode rays are not deflected by electrically charged metal plates, which would seem to indicate (incorrectly) that cathode rays cannot be charged particles.ġ886 Eugen Goldstein observes that a cathode-ray tube produces, in addition to the cathode ray, radiation that travels in the opposite direction - away from the anode these rays are called canal rays because of holes (canals) bored in the cathode later these will be found to be ions that have had electrons stripped in producing the cathode ray.ġ890 Arthur Schuster calculates the ratio of charge to mass of the particles making up cathode rays (today known as electrons) by measuring the magnetic deflection of cathode rays. James Clerk Maxwell had recognized this method soon after Faraday had published, but he did not accept the idea that electricity is composed of particles.) Stoney also proposes the name "electrine" for the unit of charge on a hydrogen ion. (He used the Faraday constant (total electric charge per mole of univalent atoms) divided by Avogadro's Number.

Crookes proposes that they are molecules that have picked up a negative charge from the cathode and are repelled by it.ġ874 George Johnstone Stoney estimates the charge of the then unknown electron to be about 10 -20 coulomb, close to the modern value of 1.6021892 x 10 -19 coulomb. Varley is first to publish suggestion that cathode rays are composed of particles. Establishes that "rays" from the cathode travel in straight lines.ġ871 C.F. Hittorf finds that a solid body put in front of the cathode cuts off the glow from the walls of the tube. He also finds that there is an extended glow on the walls of the tube and that this glow is affected by an external magnetic field.ġ869 J.W. Plücker uses Geissler tubes to show that at lower pressure, the Faraday dark space grows larger. Sprengel improves the Geissler vacuum pump. 1855 German inventor Heinrich Geissler develops mercury pump - produces first good vacuum tubes, these tubes, as modified by Sir William Crookes, become the first to produce cathode rays, leading eventually to the discovery of the electron (and a bit farther down the road to television).ġ858 Julius Plücker shows that cathode rays bend under the influence of a magnet suggesting that they are connected in some way this leads in 1897 to discovery that cathode rays are composed of electrons.ġ865 H.
